Can we solve common issues of cancer treatments through the use of a nano-sized crystal?
Common cancer treatments today can be incredibly toxic: some are insoluble in water and therefore require higher doses, and some are non-specific – affecting more than just the unhealthy areas of the body. Treatments also tend to not last very long and require patients to undergo numerous injections every week.
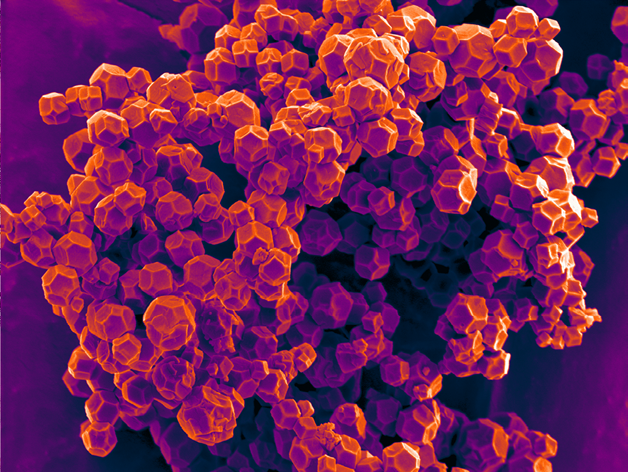
Over the years, researchers have engineered potential solutions to these problems, aiming to create solutions with slow and controlled drug delivery. Slow release allows for fewer and less frequent injections, while controlled delivery assists with less toxic side effects. Various advanced materials (some that mimic nature) are used, including polymers, peptides, and viruses. In my group, we have utilised nano-sized cages (called metal-organic frameworks) to act as both a carrier and protector, and traffic therapeutic molecules into cells.
Metal-organic frameworks are made up of two components: an organic linker and a metal cluster. They can be built from a variety of different molecule combinations. Metals that we use are biologically compatible, such as zirconium. The material chemistry is easily tuneable, and we can functionalise these materials to be targeted to specific cell receptors. This decreases off-targeting side effects and improves efficiency of treatment. We can also design the metal-organic framework to have large porosity, which is useful for encapsulating big biologic molecules compared with smaller synthetic drugs. These biologic macromolecules offer advantages over small drugs, especially related to specificity; therefore, designing a material to accommodate them offers much potential.
My research focuses on the design of these nano-sized cages to extend a drug’s efficacy – or potency – and encapsulation of a newer form of therapy (small interfering RNA) into the framework. This newer form of therapy is involved in a cellular mechanism called RNA interference, and has the ability to decrease the expression of a gene of interest. For cancer applications, this is incredibly useful and a very powerful technique. If one can limit the expression of a growth gene, for instance, it is much easier to remove a tumour before it gets too large and becomes malignant.
The potential for this research as a platform technology is wide ranging. We hope that in some years’ time, we can engineer a better solution for patients that is longer lasting, has less side effects, and involves zero reliance on patient compliance.
Michelle Teplensky
NanoDTC PhD Associate 2016